Photo credit: Pamelia Lim
Positioned at the pulmonary mucosal barrier, alveolar macrophages are often the first cells to take up inhaled pathogens, performing a critical function as immune sentinels of the lung. We study the role of alveolar macrophages during Mycobacterium tuberculosis (Mtb) infection, a respiratory pathogen that kills almost 1.5 million people each year and is the leading cause of infection-related death worldwide. Alveolar macrophages play a unique role during Mtb infection, because they are the very first cell to become infected after aerosol transmission and remain the dominant cell type infected through the first 10 days. Therefore, the speed and quality of the response of these macrophages influence disease outcome.

Role of myeloid Nrf2 expression during Mtb infection
Our previous work found that alveolar macrophages initially respond to Mtb in a non-inflammatory manner, which is dependent on expression of the transcription factor Nrf2. Nrf2 is a master regulator for an antioxidant/oxidative stress response that regulates pathways such as glutathione metabolism and antioxidant production. Mtb-infected cells must eventually initiate innate cell recruitment and priming of the adaptive response, and the molecular basis for these events remains poorly understood. Using Nrf2 conditional knock-out strains and Nrf2 chemical agonists and antagonists, we aim to characterize how the early induction of a cell protective program by Nrf2 prevents alveolar macrophages from mounting a pro-inflammatory response to Mtb infection including antigen presentation, delaying subsequent immune events, and leading to impaired bacterial control. Regulation of pulmonary inflammation by Nrf2 is also applicable to other respiratory infections and inflammatory conditions.
Alveolar macrophages are innate sensors and inflammatory mediators
We are interested in understanding how alveolar macrophages maintain dual functions within the pulmonary airway: 1) as innate sensors, recognizing Pathogen Associated Molecular Patterns (PAMPs) from direct infection, 2) as inflammatory mediators, responding to inflammatory signals in the environment derived from other responding cells. We would like to know how these different roles help alveolar macrophages to maintain pulmonary homeostasis, allowing for sufficient immune responses to infection while preventing unnecessary pathology and lung damage.
Photo credit: Greg Olson/Alissa Rothchild
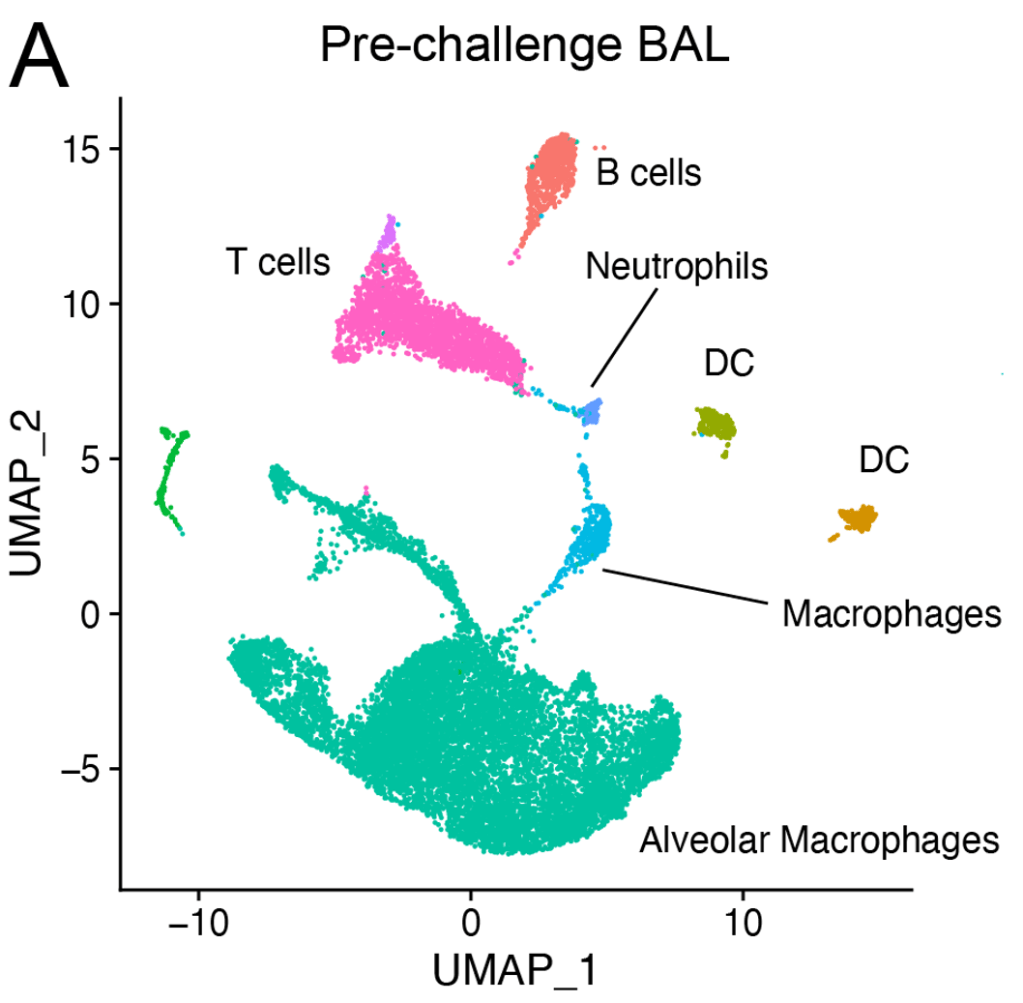
Remodeling of alveolar macrophages by vaccination and prior infection
We previously showed that alveolar macrophages mount a cell-protective Nrf2-dependent response to Mtb infection over the first ten days of infection. We are now interrogating how different inflammatory environments alter the response of alveolar macrophages and the characteristics of their plasticity, using BCG vaccination and contained Mtb infection as models of systemic inflammation. Which inflammatory signals are responsible for alveolar macrophage remodeling? How durable are those changes? What are the consequences for the host response?

These studies are part of the Cascade IMPAc-TB (Immune Mechanisms of Protection Against Mycobacterium tuberculosis) consortium, studying effective host immune responses across small animal models, non-human primates, and human cohorts.